HUMAN BRAIN EVOLUTION: WHAT ENDOCASTS FROM FOSSILIZED SKULLS REVEAL
Dean Falk; June, 2025
Hominin paleoneurologists are scientists who investigate brain evolution by studying the skulls of early human ancestors. How, you might wonder, can one learn about the evolution of the cognitive traits that define humans (language, for example) from the fossil record? This kind of research requires interpretation of details that are, literally, imprinted in stone, and that are often murky and difficult to decipher. Partly for this reason, paleoneurologists argue passionately about how, when, and why human brains evolved to their present-day state.
The evidence that paleoneurologists interpret comes mainly from the fossilized braincases of early human ancestors or their close relatives who were bipedal, known as hominins. (Modern humans are also hominins.) Traditionally, the volume of the braincase (its cranial capacity measured in cubic centimeters, cm3) has been used as a surrogate for brain size even, although the cranial capacity of a skull is somewhat of an overestimate for brain size because braincases contain fluids and membranes in addition to brains. Nevertheless, cranial capacities are easy to measure and provide an excellent window into the evolution of brain size when they are plotted against time on a graph. From such data, we know that brain size increased over many millions of years in a wide variety of mammals, including those from different branches of our own group, the primates.
Compared to most other animals (whales and elephants are an exception), humans have very large brains in absolute terms, by far the largest of any primate. The plot of absolute cranial capacities over time is shown below (Fig. 1). It reveals that average brain size in living people (around 1350-1400 cm3) is three to four times that of early human relatives called australopithecines who lived in Africa long ago. Natural selection, thus, favored an upward trend in hominin brain size during the last 3.5 million years. Some scientists associate this trend with advances in tool production and/or the evolution of neurological wiring that underpins advanced abilities like language, music, and mathematics.
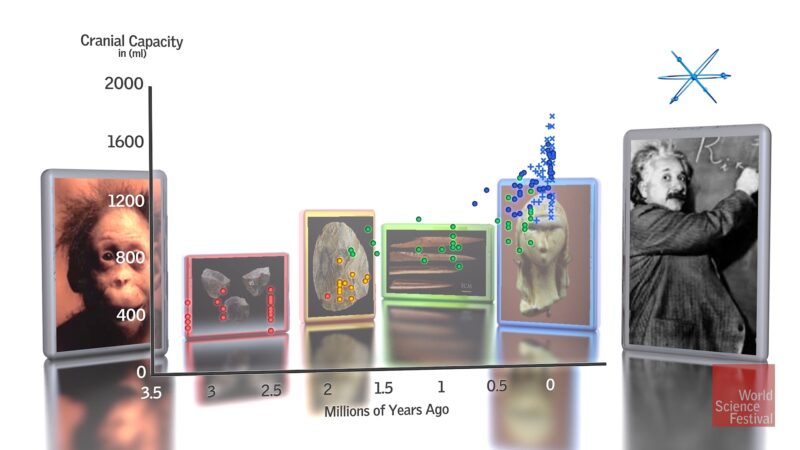
Figure 1. Cranial capacity, a surrogate for brain size, more than tripled in hominins during the last 3.5 million years (illustration from Falk, 2016).
Another trait that paleoneurologists pay attention to is the ratio of brain size to body size, which is called relative brain size (or RBS). RBS is remarkably high in humans compared to nonhuman primates or other mammals with body sizes mathematically projected to match those of humans. As an example, a little squirrel monkey with a tiny brain has the same RBS as humans, but when one mathematically enlarges the monkey to human body size, its RBS drops way below the human value. Such “allometric scaling” is why the heads of human babies appear relatively large for their bodies compared to adults. In other words, as the baby grows into an adult, both its relative head size and RBS decrease to those of adult humans (see Falk, 2022, for further discussion about allometry).
A comparison of brain development in modern species strongly suggests that brain size evolution in the human lineage depended on faster rates of brain growth in newborns and young infants compared to other primates (i.e., “brain spurts” indicated by an arrow within the shaded column in Fig. 2). It’s not just that human brains grow at an especially quick pace during the first year(s) of life, the brain’s internal wiring and neurochemistry also develop extremely rapidly as babies mature and adjust to the world. Studying and comparing the development of bodies as well as brains in humans and their closest nonhuman cousins, the great apes (especially chimpanzees), is an important way to formulate ideas about hominin evolution (Falk, 2025b).

Figure 2. Brain spurts in humans and chimpanzees. This chart shows brain size in humans of different ages compared to chimpanzees. B, birth. The shaded bar highlights the steep brain growth during the first year of life (brain spurt) for both species. The human brain spurt is steeper and, thus, facilitates a larger average adult brain size in humans compared to chimpanzees. The hominin is the Taung specimen, who died when she was less than four years old (Falk, 2025b). Reproduced from Falk, 2016.
Interestingly, it appears that brain size may have leveled off in our species, and even declined in some modern groups compared to our late relatives, the Neanderthals. As with computers, size is not everything when it comes to brains—the quality and combination of the brain’s various regions, neurochemicals, and connections is also extremely important.
Some of the most telling information about the brains of early hominins comes from internal molds of fossilized braincases, known as endocranial casts or endocasts. An endocast, such as the one from Taung shown in Figs. 2 and 3, reproduces the shape and, with luck, some of the details of the external surface of the brain (the cerebral cortex) that were imprinted on the walls of the braincase when the individual was alive (Labra et al., 2023). This is fortunate for paleoneurologists because the human cerebral cortex is the part of the brain that facilitates highly evolved functions such as language and rational problem solving. This is one reason why paleoneurologists spend a good deal of time scrutinizing endocasts from early human relatives, some of which lived several million years ago (Falk, 2014; Falk and Marom, 2024; Gunz et al., 2020). Historically, the extent to which the back end of the endocast appears apelike or humanlike in australopithecines has been contentious since 1925 when this part of the endocast was first described for Taung (Australopithecus africanus). A significant contribution was made to this debate when the back end of an australopithecine endocast from another species (Australopithecus afarensis) was demonstrated to have an apelike appearance (Gunz et al., 2020).
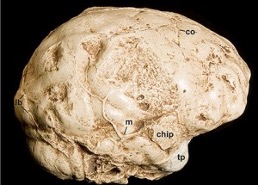
Figure 3. A nearly complete half endocast that provides information about the right side of the cerebral cortex of Taung (Australopithecus africanus). The back end of the endocast is to the left. Abbreviations: chip, chip of bone that adhered to endocast; co, coronal suture; lb, lambdoid suture; m, meningeal blood vessel; tp, temporal pole
As illustrated in Fig. 4, brains are composed of different lobes that, broadly speaking, facilitate different basic functions. For example, vision is processed in the occipital lobes at the back of the brain. Brains also have right-left differences, and these are particularly marked in humans—both visually (brains appear lopsided when viewed from the top) and functionally (in the majority of people, language is processed mostly by the left side of the brain, while music depends more on the right side). The regions between the primary cortices that represent the basic functions such as hearing, seeing, and so on are called association cortices. Association cortices synthesize and analyze information they receive from other parts of the brain (including primary cortices) and are thought to have been of paramount importance for the evolution of advanced cognition in humans.
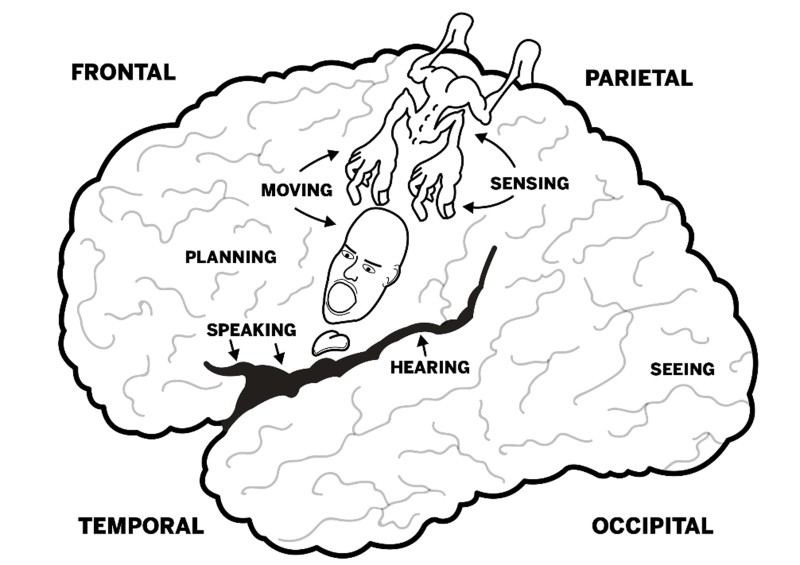
Figure 4. Lobes of the human brain and locations for some basic functions (left side of brain). The little person (homunculus) illustrates the locations that facilitate movement and reception of sensory information for specific body parts (mostly on the opposite side of the body).
The cerebral cortex of most primates (and, indeed, many other mammals) consists of convolutions of gray matter (gyri) that are separated by grooves (sulci). The patterns of sulci (sulcal patterns) can be informative, and, for this reason, paleoneurologists are keenly interested in the traces of sulci that are sometimes reproduced on endocasts from the skulls of early hominins. Genetically speaking, chimpanzees (Pan) are the closest living “cousins” of people (Homo). It is, therefore, interesting to compare the sulcal patterns that are typical for these two groups to get an idea of the changes that must have occurred at some point in the cerebral cortices of our ancestors. Toward that end, sulcal patterns have been described in detail from magnetic resonance images of the brains of 8 living chimpanzees (Fig. 5) (Falk et al. 2018).

Figure 5. Left hemispheres imaged from the brains of eight living chimpanzees, with sulci labeled. Images like these are useful to scientists who attempt to identify sulci on endocasts from fossil hominins. Reproduced from Falk et al., 2018.
The patterns of the main (named) sulci of humans do not differ very much from those of chimpanzees and the other great apes. In fact, there are only two places in the brain where the typical sulcal pattern of apes and humans differ significantly. The first is in the frontal lobes, which in apes have large curved (fronto-orbital) sulci in front of Brodmann’s area (BA) 44 that are lacking in human brains, which instead have two little sulci that form two sides of a triangular patch of cortex (BA 45). (See Fig. 6.) On the left side of the human brain, this patch is part of Broca’s speech area. The second place in the brain where apes and people are obviously different is at the back. In apes and most monkeys, a large crescent-shaped sulcus (the Affenspalte or lunate sulcus) courses just in front of the primary visual cortex (BA 17). In humans, BA 17 is shifted posteriorly around the back end of the brain onto the internal surface of the hemisphere so that, unlike apes, most of it is not seen in lateral views. The small portion that is sometimes visible on the lateral surface (as in the following illustration) is no longer bordered by a lunate sulcus, which was lost during hominin evolution (Falk, Zollikofer & Ponce de León, 2025).

Figure 6. Typical sulcal patterns of chimpanzees and humans (left sides of brain). The fronto-orbital sulcus and the lunate sulcus are red in the chimpanzee, as are the two little sulci that border Brodmann’s area 45 in the human brain. Numbers represent Brodmann’s areas (BA). BA 17 is primary visual cortex, whereas BA 44 and 45 form Broca’s speech area on the left side of human brains. Notice that BA 17 of the human is not bordered by a lunate sulcus.
WHAT HAPPENED TO THE LUNATE SULCUS? –A HYPOTHESIS
As noted, the extent to which the back end of the brain appeared apelike or humanlike in early hominins has been controversial since Raymond Dart first described Taung’s endocast in 1925. Specifically, the debate has focused on whether australopithecines had a lunate sulcus in an anterior “apelike” or posterior “humanlike” position (Fig. 6) and the implications of its location (wherever it was) for understanding the evolution of the parietotemporo-occipital association cortices that border the front of this sulcus in apes (for a historical overview, see Falk, Zollikofer, and Ponce de León, 2025). Those who claimed australopithecines had a lunate sulcus in a posterior humanlike position (as Dart had) theorized that the lunate sulcus and BA 17 were, together, “pushed” posteriorly by evolutionary expansion of the association cortices in front of them. However, a landmark study published over 80 years after Dart’s publication, showed that human brains do not have lunate sulci like those of apes, which had been a basic assumption underlying the “push-back” hypothesis (Allen, Bruss, and Damasio, 2006). This raised the question of what happened to the lunate sulcus if it, indeed, had not been pushed toward the back of the brain.
Louis Pierre Gratiolet’s insights from 1854, along with more contemporary research (Falk et al., 2018; Mangin et al., 2019), prompted recent reconsideration of the fate of the lunate sulcus in early hominins (Falk, Zollikofer, and Ponce de León, 2025). Gratiolet (1854) described folds that are buried within the depths of the lunate sulci of primates where they bridge between the parietal and occipital cortices, which he named “plis de passage” (passage folds). Today, these buried connecting folds, called annectant gyri, are known to exist within sulci distributed across the human cerebral cortex (see Mangin et al., 2019 for a review). Theoretically, some annectant gyri are thought to expand upwards while the brain is developing, where they spill over onto the cortical surface, thus, giving rise to convolutions on the surface of the brain (Magin, 2019). If so, little gyri in the depths of sulci may be thought of as incubators for the development of the brain’s external convolutions.
It’s important to note that the lunate sulcus one sees on the brains of apes and monkeys is only the superficial edge of the sulcus, which actually consists of a deeper slit or pocket that separates the parietal (and temporal) lobes from the occipital lobe. In other words, the superficially visible lunate sulcus is at the top of the whole sulcus and is, thus, said to cover it up (or “opercularize” it –operculum means “lid”). Based on Gratiolet (1854), Magin et al. (2019), and our research on chimpanzees and humans (Falk et al., 2018; Falk, Zollikofer, and Ponce de León, 2025), we propose that, rather than being “pushed” posteriorly during hominin evolution, the lunate sulcus was a hotspot for expansion of annectant gyri onto the cortical surface where they gave rise to the external convolutions of the occipital cortex. Zollikofer coined the term “de-opercularization” for this evolutionary process, which I visualize as much like turning a pocket on a pair of pants inside out (Fig. 7). In so doing, the pocket (deep, hidden part of the lunate sulcus) and its external opening (C-shaped part of the sulcus that is visible on the surface of the brain) no longer exist. Instead, there’s material overlying where they used to be (occipital convolutions)!

Figure 7. Falk, Zollikofer, and Ponce de León (2025) hypothesize that, similar to turning a pants pocket inside out, the lunate sulcus was lost during hominin evolution when connecting gyri within its depths expanded upward and overspilled onto the cortical surface, thus giving birth to the external gyri that connect the parietal & occipital lobes.
WHEN DID THE LUNATE SULCUS BEGIN TO DE-OPERCULARIZE?
Falk, Zollikofer, and Ponce de León (2025) explored the possible timing of evolutionary changes in the lunate sulcus by analyzing the parietooccipital region on virtual endocasts from four early Homo adults from Dmanisi, Georgia, dated to ~ 1.8 million years ago. Their results suggest that annectant gyri were beginning to emerge onto the outside of the brain, fragmenting the lunate sulcus in the process as the Dmanisi population was evolving larger brains with more backward-projecting occipital lobes compared to earlier hominins. This finding is consistent with earlier research on the frontal lobes of the Dmanisi endocasts, which concluded that these early hominins may have ushered in a relatively narrow period of time during which the external cortical surface was beginning to appear more humanlike than apelike (Ponce de León et al., 2021).
Although, with luck, endocasts may tell us something about the outside surface of the brain, they are notoriously uninformative when it comes to understanding its internal anatomy, including the neurological connections between various parts (wiring). However, since annectant gyri are buried within the depths of sulci (Gratiolet, 1854; Mangin et al., 2019), the apparent partial emergence of well-known annectant gyri onto the surface of the brain in the Dmanisi early hominins suggests that neurological reorganization may, more generally, begin evolving inside the brain before being manifested in evolved patterns of cortical gyri and sulci on its external surface.
BEYOND ENDOCASTS
Traditionally, evolutionary anthropologists study the emergence of advanced cognitive abilities by focusing on the material culture, especially stone tools, that our ancestors deliberately manufactured, starting around 3.5 million years ago (Falk, 2025c). The span of time between the first appearance of stone tools and the beginning of the Metal Ages around 6,000 years ago is widely recognized as the Stone Age (Fig. 8). Some researchers believe a first “major leap” in hominin brain evolution occurred when hominins began to manufacture particularly refined stone tools (Acheulean hand axes) around 2.0 million years ago in conjunction with the beginning of an upward turn in the volumes of cranial capacities (surrogates for brain size) (Fig. 1). Others suggest that major cognitive advances did not occur until much more recently, perhaps in association with language evolution and/or refinement of artistic abilities.
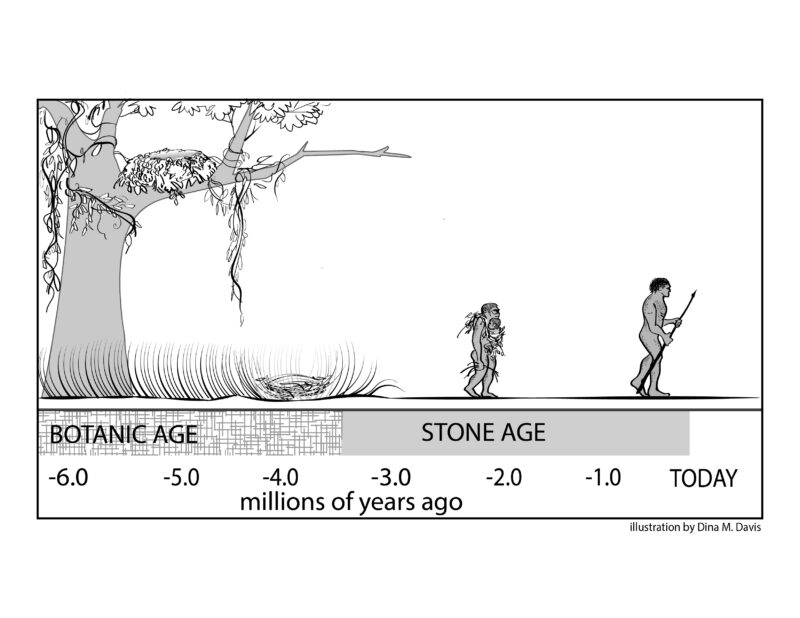
Figure 8. The duration of the Stone Age lasted from the time stone tools first appeared in the archaeological record (~ 3.5 million years ago) to the beginning of the Metal Ages (~ 6,000 year ago). Illustration by Dina Davis, reproduced from Falk, 2025c.
Although these ideas are prevalent in textbooks, they miss an important point. Extensive evidence shows that hominin ancestors split from those of our cousins, the chimpanzees, some 5-7 million years ago (Fig. 9). Since the earliest known tools are just under 3.5 million years old (~ the beginning of the Stone Age), early hominins appear to have been evolving for 1.5 to 3.5 million years before they ever knapped a rock into a useful tool.
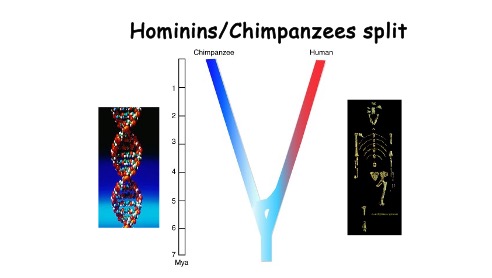
Figure 9. Extensive genetic and fossil evidence indicates that the ancestors of chimpanzees and the hominin predecessors of humans split into two lineages between 5-7 million years ago. The fossil hominin is that of the famous ‘Lucy’ (Australopithecus afarensis), dated to ~ 3.2 million years ago.
Elsewhere I have dubbed the approximately first half of hominin evolution the Botanic Age (Fig. 8; Falk, 2025c) and used comparative evidence to argue that the hominins from that mysterious time evolved in ways that seeded the later emergence of abilities that underpinned the vastly more recent evolution of contemporary language(s), music, mathematics, and art (Falk, 2025a; Larsson and Falk, 2025). In short, I speculate that hominins spent their “formative years” in the Botanic Age and, further, that natural selection (or Mother Nature) acted on mothers and infants in ways that started hominins meandering down a path that led, for better or worse, to humanity as we know it.
REFERENCES
Allen, J. S., J. Bruss, and H. Damasio. 2006. Looking for the lunate sulcus: a magnetic resonance imaging study in modern humans. Anat Rec A Discov Mol Cell Evol Biol 288 (8):867-76.
Dart, R. A. 1925. Australopithecus africansus: The man-ape of South Africa. Nature 115:195-199.
Falk, D. 2022. Implications of brain evolution in Cetaceans and Primates for highly intelligent extraterrestrial life. Journal of Astrobiology 12:45-64.
Falk, D. 2025a. Don’t ignore cognitive evolution during the three million years that preceded the archaeological record of material culture! Behavioral and Brain Sciences: 48:e8. https://doi.org/10.1017/S0140525X2400102X
Falk, D. 2025b. ‘Taung Child’ fossil offers clues about the evolution of childhood. Nature 638:29-31.
Falk, Dean. 2025c. The Botanic Age, Planting the Seeds of Human Evolution. Toronto University of Toronto Press.
Falk, D. 2016. Evolution of brain and culture: The neurological and cognitive journey from Australopithecus to Albert Einstein, Journal of Anthropological Sciences 94:1-14. https://pubmed.ncbi.nlm.nih.gov/26894688/
Falk, D. 2014.Interpreting sulci on hominin endocasts: old hypotheses and new findings. Frontiers in Human Neuroscience 8:134. https://doi.org/10.3389/fnhum.2014.00134
Falk, D. & A. Marom. 2024. The DNH7 endocast of Paranthropus robustus from Drimolen, South Africa: Reconsidering the functional significance of an enlarged occipital-marginal (O/M) sinus system in robust australopithecines. American Journal of Biological Anthropology: e25010:1-10. https://onlinelibrary.wiley.com/doi/pdf/10.1002/ajpa.25010
Falk, D., Zollikofer, C. P. E., M. S. Ponce de León. 2025. De-opercularization of the lunate sulcus in early Homo. The Anatomical Record, 1-19. https://doi.org/10.1002/ar.25694
Falk, D., C.P.E. Zollikofer, M. Ponce de León, K. Semendeferi, J.L. Alatorre Warren, W.D. Hopkins. 2018. Identification of in vivo Sulci on the external surface of eight adult chimpanzee brains: Implications for interpreting early hominin endocasts. Brain, Behavior and Evolution. https://doi.org/10.1159/000487248
Gratiolet, Louis Pierre. 1854. Mémoire sur les plis cérébraux de l’homme et des primatès: Mit einem Atlas (4 pp. XIV pl.) in fol. 33i. Paris: A. Bertrand.
Gunz, P., S. Neubauer, D. Falk, P. Tafforeau, A. Le Cabec, T.M. Smith, W.H. Kimbel, F. Spoor, Z. Alemseged. 2020. Australopithecus afarensis endocasts suggest ape-like brain organization and prolonged brain growth. Science Advances 6 (14), April 1. https://advances.sciencemag.org/content/6/14/eaaz4729
Labra, Nicole, Yann Leprince, Denis Rivière, Mathieu Santin, Jean François Mangin, Lou Albessard-Ball, Amélie Beaudet, Emiliano Bruner, Douglas Broadfield, Kristian Carlson, Zachary Cofran, Dean Falk, Emmanuel Gilissen, Aida Gomez Robles, Aurélien Mounier, Simon Neubauer, Alannah Pearson, Carolin Röding, Yameng Zhang, Antoine Balzeau. 2023. What do brain endocasts tell us? A comparative analysis of the accuracy of sulcal identification by experts and perspectives in palaeoanthropology. Journal of Anatomy, 00, 1-23: https://doi.org/10.1111/joa.13966
Larsson, M. & D. Falk. 2025. Direct effects of bipedalism on early hominin fetuses stimulated later musical and linguistic evolution, forum article with international commentaries. Current Anthropology 66(2): 257-278.https://doi.org/10.1086/734554
Mangin, Jean-François, Yann Le Guen, Nicole Labra, Antoine Grigis, Vincent Frouin, Miguel Guevara, Clara Fischer, Denis Rivière, William D. Hopkins, Jean Régis, and Zhong Yi Sun. 2019. “Plis de passage” Deserve a Role in Models of the Cortical Folding Process. Brain Topography 32 (6):1035-1048.
Ponce de León, Marcia S, Thibault Bienvenu, Assaf Marom, Silvano Engel, Paul Tafforeau, José Luis Alatorre Warren, David Lordkipanidze, Iwan Kurniawan, Delta Bayu Murti, and Rusyad Adi Suriyanto. 2021. The primitive brain of early Homo. Science 372 (6538):165-171.

